|
Consistency
Quality Control and Testing
Product Selection & Support
HiPSC Custom Services
|
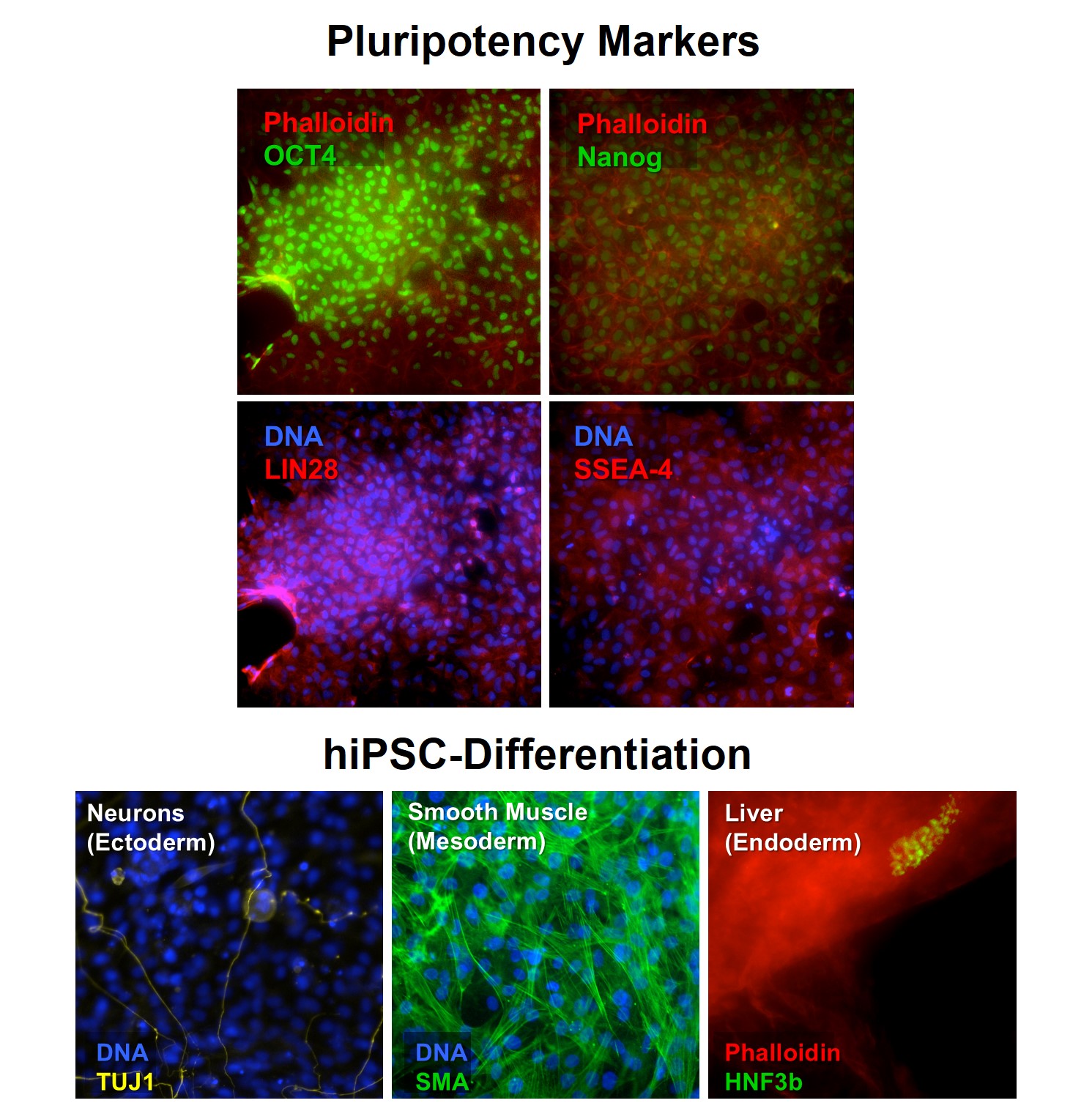 Human Induced Pluripotent Stem Cells (HiPSC) Top: HiPSC express pluriotency markers OCT4, Nanog, LIN28 and SSEA-4. Bottom: HiPSC differentiate into cell derivatives from the 3 embryonic layers: Neuronal marker beta III tubulin (TUJ1), Smooth Muscle Actin (SMA) and Hepatocyte Nuclear Factor 3 Beta (HNF3b).
|
|
Menu
|
|
Human iPSC
|
|
iPSC-Derived Cardiac Cells
|
|
iPSC-Derived Neural Stem Cells
|
|
iPSC & Derived Cell Resources
|
Cutting-edge development and manufacturing provides high quality, thoroughly-characterized HiPSC cells to researchers around the world. HiPSC are generated from somatic cells, eliminating ethical considerations associated with scientific work based on embryonic stem cells. Furthermore, being donor/patient-specific, they open possibilities for a wide variety of studies in biomedical research. Donor somatic cells carry the genetic makeup of the diseased patient, hence HiPSC can be used directly to model disease on a dish.
Thus, one of the main uses of HiPSC has been in genetic disease modeling in organs and tissues, such as the brain (Alzheimer’s, Autism Spectrum Disorders), heart (Familial Hypertrophic, Dilated, and Arrhythmogenic Right Ventricular Cardiomyopathies), and skeletal muscle (Amyotrophic Lateral Sclerosis, Spinal Muscle Atrophy). The combination of HiPSC technology and gene editing strategies such as the CRISPR/Cas9 system creates a powerful platform in which disease-causing mutations can be created “on demand” and sets of isogenic cell lines (with and without mutations) serve as convenient tools for disease modeling studies.
Other applications of HiPSC and iPSC-differentiated cells include drug screening, development, efficacy and toxicity assessment. As an example, through the FDA-backed CiPA (Comprehensive in vitro Pro-Arrhythmia Assessment) initiative, HiPSC-derived cardiac muscle cells (cardiomyocytes) are poised to constitute a new standard model for the evaluation of cardiotoxicity of new drugs, which is the main reason of drug withdrawal from the market. Finally, HiPSC-differentiated cells are being used in early stage technology development for applications in regenerative medicine. Bio-printing and tissue constructs have also been considered as attractive applications for HiPSC.
Human iPSC and Derived Cells are for Research Use Only (RUO). Not for human clinical or therapeutic use.
